^ Merck Index of Chemicals and Drugs Archived at the Wayback Machine, 14th ed.Journal of the American Chemical Society. number of negative charges so that the compound has no charge overall. While these are the most common valences, the real behavior of electrons is less simple. The usual charge of an element is common to its group. You may assume that the valences of the elementsthe number of electrons with which an atom will bond or formare those that can be derived by looking at the groups (columns) of the periodic table. 
for an unconscious person bleeding from the head. According to the Gastonia Police Department, officers were called to a home on Fraley Church Road near Smyre Drive just before 5:30 p.m. "The Thermal Decomposition of Silver Oxide". There are four ways to find the charge of an element: Use the periodic table. (WBTV) - Police have arrested a suspect accused of killing a man with a toilet tank lid inside a Gastonia home on Thursday evening. ^ General Chemistry by Linus Pauling, 1970 Dover ed.zinc iron (II) iron (III) gallium silver. ^ US 20050050990A1, Harigae, Kenichi & Shoji, Yoshiyuki, "Fine-grain silver oxide powder", published box is the intersection between the zinc cation and the chloride anion, so you should write ZnCl2, as shown.A Critical Survey of the Solubility Equilibria of Ag 2O". You can discover more on the implications of the anion gap below the form. An electrostatic attraction between anions Question 6 The reason that a sodium atom bonds with a chlorine atom is. But when we pour those two solutions together, we form our precipitate. "Studies on the Hydrolysis of Metal Ions. This anion gap calculator allows you to compute the AG value and delta gap for the sodium, chloride and bicarbonate values you input in the Na Cl and HCO3 fields. An ion has a positive or negative charge. So those water molecules are stabilizing the chloride anion in solution. ^ Biedermann, George Sillén, Lars Gunnar (1960).The Ag 4d electrons are fully included using an. "Inorganic Chemistry" Academic Press: San Diego, 2001. lattice parameter, elastic constants, TO frequency, band structure, and valence charge distribution of AgCl. This reaction does not afford appreciable amounts of silver hydroxide due to the favorable energetics for the following reaction: 2 AgOH ⟶ Ag 2 O + H 2 O : CS1 maint: multiple names: authors list ( link) Collective Volume, vol. 4, p. 547 Silver oxide can be prepared by combining aqueous solutions of silver nitrate and an alkali hydroxide. 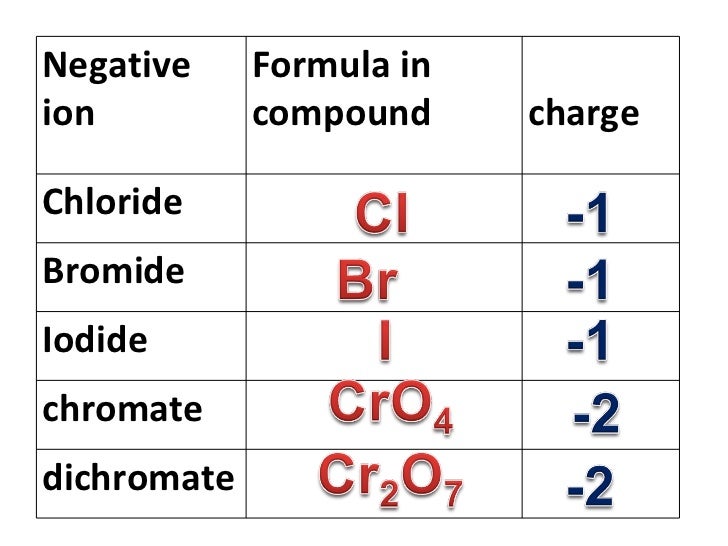
Silver(I) oxide produced by reacting lithium hydroxide with a very dilute silver nitrate solution 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
